

A review on the application of lithium isotopes in continental weathering research and progress
Received date: 2024-05-20
Revised date: 2024-10-22
Online published: 2025-02-21
Supported by
Open Fund Key Project of Jinhua Key Laboratory of Watershed Earth Surface Processes and Ecological Security(KF-2022-04)
National Natural Science Foundation of China(41572345)
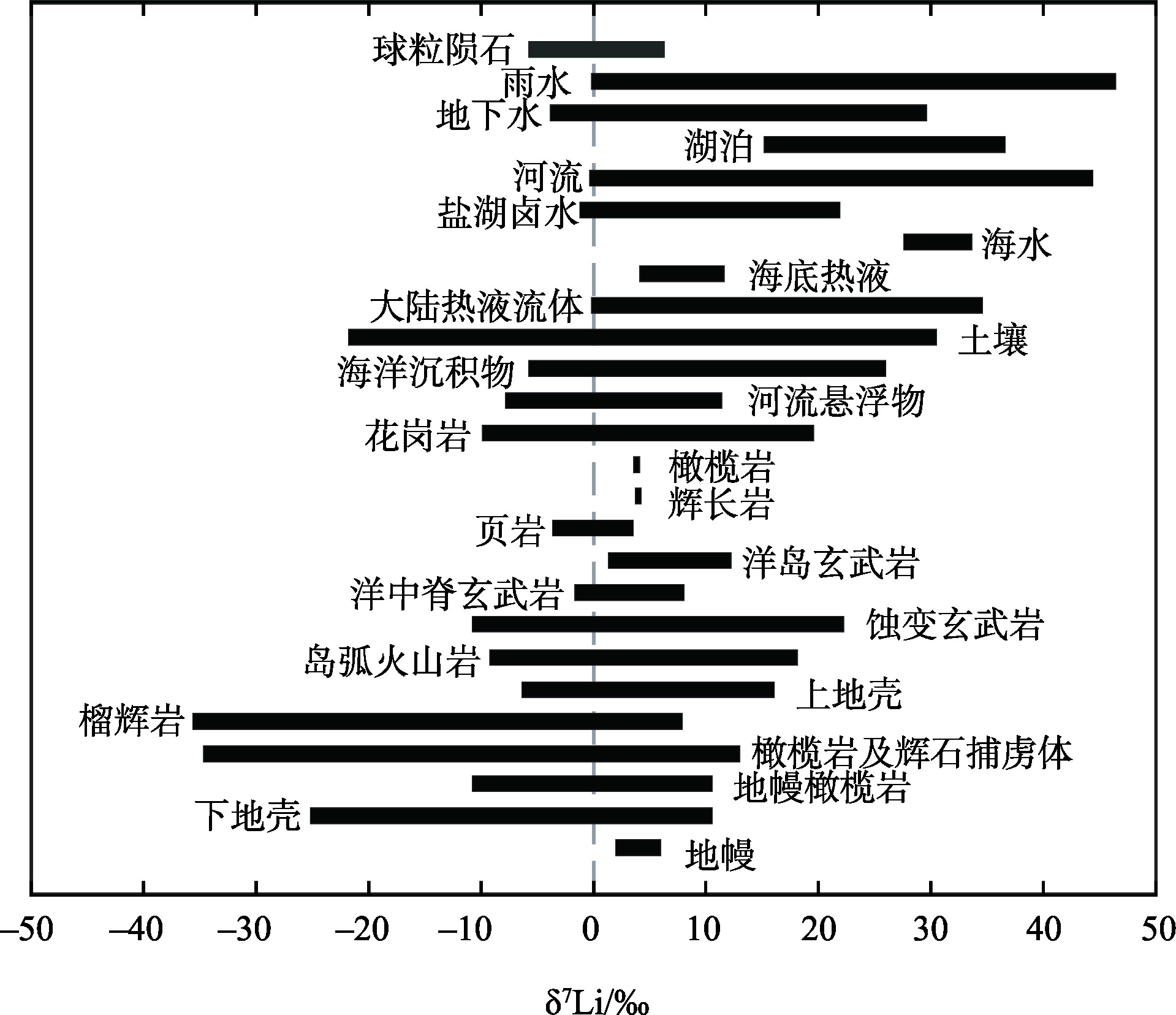
Continental chemical weathering is one of the key processes shaping the Earth's surface morphology, influencing the global material cycle and regulating the global climate. Effectively tracing surface weathering processes has consistently been a focal point within geosciences. Lithium and its isotopes, distinguished by their unique chemical properties, exhibit great potential in tracing continental silicate weathering. This article systematically reviewed the literature and analyzed the fractionation behavior and patterns of lithium isotopes during chemical weathering processes in experimental simulations, weathering profiles, and at the watershed scales. Based on this analysis, the following insights are obtained: 1) The continental weathering process in surface environments is complex, exhibiting a nonlinear relationship between chemical weathering intensity and the δ7Li value. 2) Integrative studies of published data indicate that the relationship between the δ7Li values of weathered materials and lithium content in highly weathered regions exhibits two patterns (The variation in Li content is limited, yet the δ7Li values exhibit significant differentiation; conversely, there is a certain degree of variation in Li content when the differentiation of δ7Li values is limited), with the underlying causes requiring further investigation. 3) The red earth region in southern China is an ideal area for weathering research, showing potential in exploring the mechanisms and patterns of lithium isotope fractionation. This article argues that there are still issues in using lithium isotopes as a tracer for chemical weathering, such as insufficient quantitative studies of complex processes, limited multi-scale integrated research, unclear mechanisms of lithium isotopes in highly weathered products, and the lack of a global weathering estimation model. Future efforts should focus on enhancing laboratory simulations, empirical studies, and multi-scale integrated research to further explore the potential of lithium isotope mechanisms in underexplored areas and validate the effectiveness of lithium isotope tracing in chemical weathering through the combined use of multi-isotope systems.
ZHU Lidong , WANG Ji , YU Ruifei , LI Fengquan , YOU Yijing , LU Haixin . A review on the application of lithium isotopes in continental weathering research and progress[J]. PROGRESS IN GEOGRAPHY, 2025 , 44(2) : 211 -225 . DOI: 10.18306/dlkxjz.2025.02.001
图5 高风化地区基岩风化壳、红土、黄土等地质储库的δ7Li值与Li含量图注:数据引自文献[28,34-35,46,49,59,73,75,77,82,91,114];QJ、PJLK、TX、JL为课题组未发表数据;HG为广东雷琼玄武岩风化壳样品,由于其δ7Li值与Li含量之间的关系与文中所述的分布模式不符,因此未在文中列出。 Fig.5 δ7Li value versus lithium content of geological reservoirs such as weathered crust of basic rock and red earth and loess in high weathering area |
| [1] |
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
|
| [6] |
蒋浩, 徐志方, 赵童, 等. 青藏高原流域岩石风化速率及其控制机制: 以贡嘎山地区典型地质背景小流域研究为例[J]. 第四纪研究, 2018, 38(1): 278-286.
[
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
孙明照. 硅酸盐风化速率及其控制因素: 来自不同“小流域系统”的研究[D]. 南京: 南京大学, 2018.
[
|
| [20] |
|
| [21] |
|
| [22] |
顾兆炎, 丁仲礼, 熊尚发, 等. 灵台红粘土和黄土—古土壤序列的地球化学演化[J]. 第四纪研究, 1999, 19(4): 357-365.
[
|
| [23] |
尹秋珍, 郭正堂. 中国南方的网纹红土与东亚季风的异常强盛期[J]. 科学通报, 2006, 51(2): 186-193.
[
|
| [24] |
熊文婷, 朱丽东, 张晓, 等. 中国南方红土记录的中更新世气候转型及其环境效应[J]. 第四纪研究, 2020, 40(6): 1441-1452.
[
|
| [25] |
靳华龙, 万世明. 新生代气候变冷机制研究进展[J]. 海洋地质与第四纪地质, 2019, 39(5): 71-86.
[
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
刘英俊. 元素地球化学[M]. 北京: 科学出版社, 1984.
[
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
陈瑜, 徐飞, 程宏飞, 等. 锂同位素地球化学研究新进展[J]. 地学前缘, 2023, 30(5): 469-490.
[
|
| [42] |
|
| [43] |
|
| [44] |
苟龙飞, 金章东, 贺茂勇. 锂同位素示踪大陆风化: 进展与挑战[J]. 地球环境学报, 2017, 8(2): 89-102.
[
|
| [45] |
|
| [46] |
|
| [47] |
|
| [48] |
汤艳杰, 张宏福, 英基丰. 锂同位素分馏机制讨论[J]. 地球科学, 2009, 34(1): 43-55.
[
|
| [49] |
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
郑永飞, 徐宝龙, 周根陶. 矿物稳定同位素地球化学研究[J]. 地学前缘, 2000, 7(2): 299-320.
[
|
| [56] |
|
| [57] |
|
| [58] |
|
| [59] |
|
| [60] |
|
| [61] |
|
| [62] |
|
| [63] |
|
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
莫申国, 张百平, 程维明, 等. 青藏高原的主要环境效应[J]. 地理科学进展, 2004, 23(2): 88-96.
[
|
| [68] |
|
| [69] |
|
| [70] |
|
| [71] |
张俊文, 闫雅妮, 孟俊伦, 等. 大兴安岭地区花岗岩风化过程中锂同位素地球化学行为[J]. 矿物岩石地球化学通报, 2023, 42(4): 873-881.
[
|
| [72] |
|
| [73] |
|
| [74] |
张俊文. 花岗岩风化过程锂同位素行为及其环境指示意义[D]. 武汉: 中国地质大学, 2018.
[
|
| [75] |
|
| [76] |
|
| [77] |
仝凤台. 典型硅酸盐风化过程的锂同位素地球化学行为[D]. 合肥: 中国科学技术大学, 2021.
[
|
| [78] |
刘文景. 江西龙南花岗岩风化过程的元素和Sr、Li同位素地球化学特征及其指示意义研究[D]. 北京: 中国科学院大学, 2013.
[
|
| [79] |
|
| [80] |
常成. 喀斯特地区云南石林典型红色风化壳的地球化学特征[D]. 北京: 北京科技大学, 2023.
[
|
| [81] |
|
| [82] |
|
| [83] |
|
| [84] |
杨承帆, 杨守业, 苟龙飞, 等. 锂同位素示踪表生风化与环境演变: 回顾与展望[J]. 矿物岩石地球化学通报, 2018, 37(5): 841-851.
[
|
| [85] |
|
| [86] |
|
| [87] |
|
| [88] |
李德文, 崔之久, 刘耕年. 风化壳研究的现状与展望[J]. 地球学报, 2002, 23(3): 283-288.
[
|
| [89] |
|
| [90] |
|
| [91] |
|
| [92] |
|
| [93] |
|
| [94] |
|
| [95] |
|
| [96] |
|
| [97] |
|
| [98] |
|
| [99] |
|
| [100] |
|
| [101] |
|
| [102] |
张俊文, 闫雅妮, 赵志琦, 等. 青藏高原山地小河流溶解态Li同位素地球化学行为: 以尼洋河为例[J]. 中国科学: 地球科学, 2023, 53(8): 1886-1897.
[
|
| [103] |
|
| [104] |
宋以龙. 我国大型河流流域硅酸盐岩风化过程的锂同位素制约[D]. 天津: 天津大学, 2021.
[
|
| [105] |
|
| [106] |
|
| [107] |
|
| [108] |
|
| [109] |
傅寒晶, 简星, 梁杭海. 硅酸盐化学风化强度评估的沉积物指标与方法研究进展[J]. 古地理学报, 2021, 23(6): 1192-1209.
[
|
| [110] |
|
| [111] |
蔡晓娟, 季宏兵. 石林红土风化剖面锂同位素特征[J]. 金属世界, 2019(1): 42-44.
[
|
| [112] |
|
| [113] |
|
| [114] |
|
| [115] |
|
| [116] |
|
| [117] |
|
| [118] |
叶玮, 朱丽东, 李凤全, 等. 中国中亚热带网纹红土的地球化学特征与沉积环境[J]. 土壤学报, 2008, 45(3): 385-391.
[
|
| [119] |
|
| [120] |
朱丽东, 谷喜吉, 叶玮, 等. 洞庭湖周边地区第四纪红土粒度特征及环境意义[J]. 地理科学进展, 2014, 33(1): 13-22.
[
|
/
| 〈 |
|
〉 |